When?
Start:
May 20, 2026
12:00 PM EST
End:
May 20, 2026
Speakers

Anca Copaescu
CEO Clinical Maestro®
Sameer Tandon
Senior Director, R&D Strategic & Innovative Transactions, Bristol Myers Squibb

Andrei Antonescu
Head of Product, Clinical Maestro
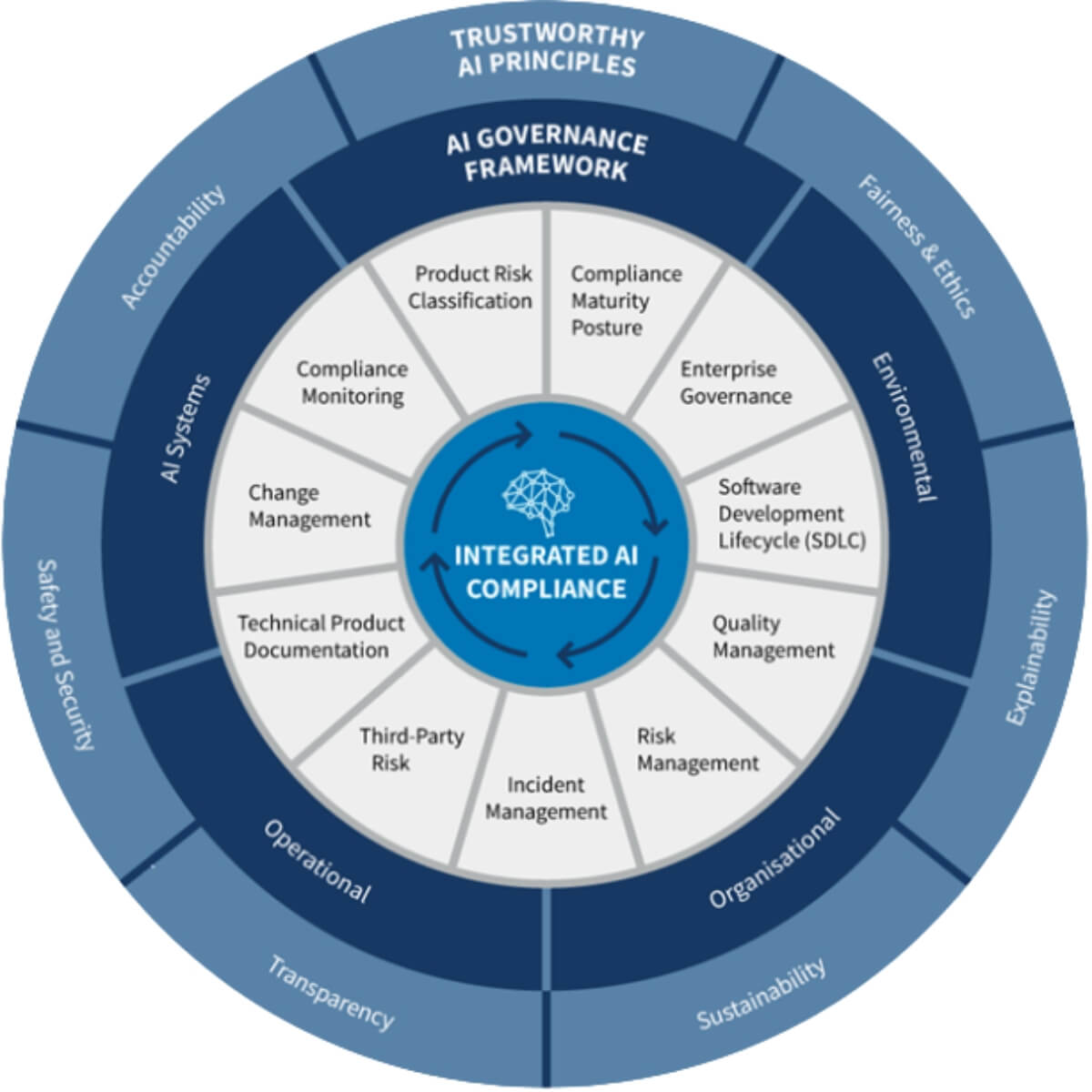
Oversight is no longer periodic or document-driven.
It must be continuous, risk-based, and fully traceable across vendors, studies, and services.
Yet most pharma organizations still operate with:
- fragmented systems
- manual workflows
- limited visibility
👉 That’s where control breaks down.
🚨 Why This Matters Now
E6(R3) does not change accountability - it changes how oversight must be demonstrated.
Sponsors are now expected to:
- continuously monitor vendor risk
- ensure decision traceability
- maintain structured governance across the lifecycle
This exposes a critical gap:
👉 Vendor management processes don’t scale to E6(R3) expectations
At this level of complexity, this is no longer a process issue - it’s a systems and orchestration challenge.
🎯 Join the Webinar
📅 20 May 2026 | 12 PM EST
📍 LinkedIn Live
A practical, experience-driven session on what vendor oversight looks like in reality — not theory.
👥 Speakers
Anca Copaescu
CEO, Clinical Maestro
Sameer Tandon
Senior Director, R&D Strategic & Innovative Transactions
Bristol Myers Squibb
Andrei Antonescu
Head of Product, Clinical Maestro
🔍 What You’ll Learn
⚠️ Risk — From Identification to Continuous Monitoring
- Why risks are recognized too late
- Limits of periodic and manual assessments
- Moving to continuous, trigger-based monitoring
🧾 Compliance — Inspection Readiness & E6(R3)
- What E6(R3) requires in practice
- From documentation to decision traceability
- Why most organizations are not fully prepared
📊 Performance — From KPIs to Decision Support
- Why traditional KPIs fail
- Connecting performance with risk and outcomes
- Moving to real-time, actionable insights
🧠 Orchestration — Connecting Everything
- Why fragmented tools don’t scale
- The need for a single source of truth
- Structured workflows and end-to-end visibility
🎯 Sponsor Perspective — Reality at Scale
- Managing vendor complexity in large pharma
- Trade-offs between speed, cost, and compliance
- What actually works — and what doesn’t
🧠 What Makes This Different
This is not a product demo.
This is not a theoretical discussion.
You will hear:
- real challenges from large pharma
- practical insights grounded in operations
- how organizations are evolving toward structured oversight
🚀 Who Should Attend
- Clinical Operations Leaders
- Vendor Management / Outsourcing Heads
- Procurement & TPRM Teams
- Quality & Compliance Leaders
- Pharma & Biopharma Executives
👉 Secure Your Spot
If you are responsible for vendor oversight in clinical trials, this session will help you understand:
- where your current approach may fall short
- what E6(R3) means operationally
- how to move toward scalable, structured oversight
Strengthen your processes and increase data-driven decision-making for faster study start-ups.
When?
Start:
May 20, 2026
12:00 PM EST
End:
May 20, 2026
Speakers

Anca Copaescu
CEO Clinical Maestro®
Sameer Tandon
Senior Director, R&D Strategic & Innovative Transactions, Bristol Myers Squibb

Andrei Antonescu
Head of Product, Clinical Maestro
Strengthen your processes and increase data-driven decision-making for faster study start-ups.
RESOURCES
Explore Expert Insights and Resources for Clinical Excellence

Webinars
Oversight is no longer periodic or document-driven. It must be continuous, risk-based, and fully traceable across vendors, studies, and services.

Blogs
Clinical Maestro® launches Clinical Maestro® 5.0 with Clinical Maestro AI — transforming clinical outsourcing. Learn how sponsors and CROs benefit from vendor intelligence, rate card compliance, change order simplification, and cost transparency.

Case Studies
Faced with inefficiencies, compliance risks, and fragmented communication, a biopharma company dramatically enhanced its vendor governance by implementing VISION!
DEMO
Request a demo
Discover how Clinical Maestro®’s advanced solutions can streamline your clinical trial operations. Request a personalized demo to explore how our tools transform budgeting, vendor management, and outsourcing efficiency for pharma and biotech.